
Regulatory Trends in Cosmetics: 12–18 Month Outlook

This guide was published on 12th November, 2025. Further regulatory developments may have occurred after publication. To keep up-to-date with the latest compliance news, sign up to our newsletter.
A practical, 12–18 month roadmap for cosmetics teams who need to move fast, without losing control of compliance.
Regulatory Trends in Cosmetics: 12–18 Month Outlook
Cosmetics requirements are shifting quickly across major markets, especially in the United States and the European Union. Staying aligned means tracking change across product safety expectations, ingredient restrictions, labeling, claims, and quality systems.
This guide gives you a clear view of the topics most likely to affect cosmetics businesses over the next 12–18 months, including where regulatory density is increasing and which themes are drawing the most attention. (In our data, cosmetics regulations have grown sharply in recent years.)
If you’re responsible for regulatory readiness, reformulation planning, labeling, or market access, this is designed to help you prioritize what to watch and what to act on first.
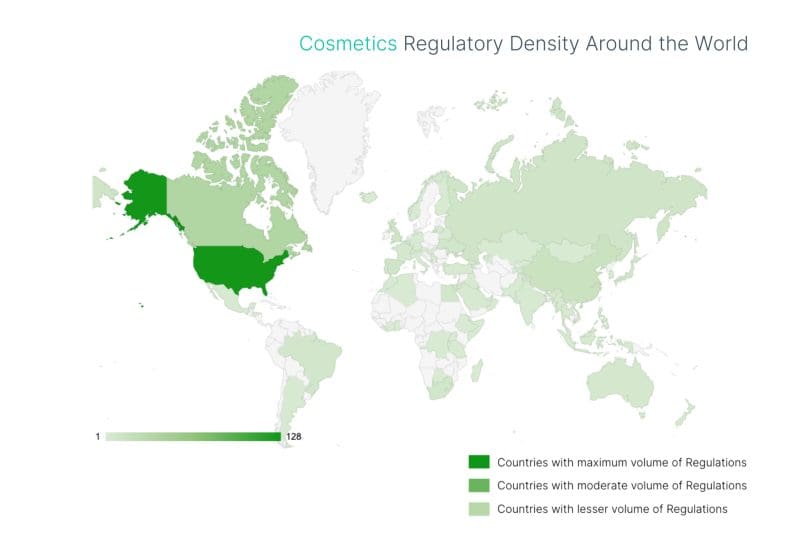
(Source: C2P by Compliance & Risks)
This Guide Covers
- Key US developments shaping cosmetics compliance, including MoCRA-driven rulemaking areas
- EU updates influencing labeling and CMR (carcinogenic, mutagenic, or reprotoxic) classification considerations
- The major topic areas to monitor over the next 12–18 months (restrictions, claims, allergens, GMP, reporting and more)
- PFAS, packaging, and traceability themes that are increasingly connected to cosmetics obligations
- Why counterfeit cosmetics remain a growing safety and compliance concern
- A curated list of key regulations referenced in the outlook








